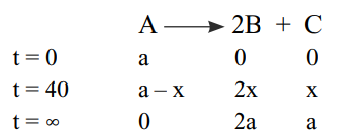- Tardigrade
- Question
- Chemistry
- When sucrose is hydrolysed, it produces dextrorotatory glucose and laevorotatory fructose whereas the reactant itself is a dextrorotatory compound. The above a conversion process follows first order kinetics. Similarly, an optically active compound A is hydrolysed as follows. A + H 2 O xrightarrow H + 2 B + C The observed rotation of compound A, B and C are 60°, 50° and -80° per mole respectively. The angles of rotation after 40 minutes and after the completion of reaction were 26° and 10° respectively. At 27° C activation energy for conversion is 27 kJ mol -1. (Use: log 1.25=0.0969, log 14.97= 1.175) The value of rate constant of the above reaction at 27° C is
Q.
When sucrose is hydrolysed, it produces dextrorotatory "glucose" and laevorotatory "fructose" whereas the reactant itself is a dextrorotatory compound. The above a conversion process follows first order kinetics. Similarly, an optically active compound A is hydrolysed as follows.
The observed rotation of compound A, B and C are , and per mole respectively. The angles of rotation after minutes and after the completion of reaction were and respectively. At activation energy for conversion is . (Use: 1.175)
The value of rate constant of the above reaction at is
Solution: