- Tardigrade
- Question
- Chemistry
- Molar solubility of helium, nitrogen and oxygen are plotted against partial pressure o f the gas at constant temperature. Henry’s law constant for these gases will lie in the following sequence? <img class=img-fluid question-image alt=image src=https://cdn.tardigrade.in/img/question/chemistry/ad30c62b3112c6bd7098c14c4bbce2f9-.png />
Q.
Molar solubility of helium, nitrogen and oxygen are plotted against partial pressure o f the gas at constant temperature.
Henry’s law constant for these gases will lie in the following sequence?
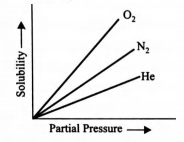
Solution:
Gas
Temperature
(K bar)
He
293
144.97
293
76.48
293
34.86
The higher the value of Henry’s law constant, the lower is the solubility of the gas in the liquid
| Gas | Temperature | (K bar) |
|---|---|---|
| He | 293 | 144.97 |
| 293 | 76.48 | |
| 293 | 34.86 |