Q.
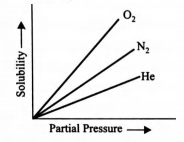
Molar solubility of helium, nitrogen and oxygen are plotted against partial pressure o f the gas at constant temperature.
Henry’s law constant for these gases will lie in the following sequence?
Solutions
Solution:
Gas
Temperature
$K_{H}$(K bar)
He
293
144.97
$N_{2}$
293
76.48
$O_{2}$
293
34.86
The higher the value of Henry’s law constant, the lower is the solubility of the gas in the liquid
| Gas | Temperature | $K_{H}$(K bar) |
|---|---|---|
| He | 293 | 144.97 |
| $N_{2}$ | 293 | 76.48 |
| $O_{2}$ | 293 | 34.86 |