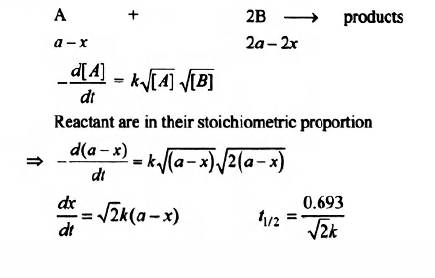Q.
For the reaction products (started with concentrations taken in stoichiometric proportion), the experimentally determined rate law is:
The half life time of the reaction would be:
Solution:
Correct answer is (c)