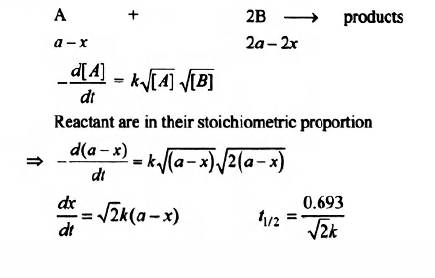Q.
For the reaction $A + 2B \to$ products (started with concentrations taken in stoichiometric proportion), the experimentally determined rate law is:
$-\frac{d\left[A\right]}{dt}=k\sqrt{\left[A\right]}\sqrt{\left[B\right]}$
The half life time of the reaction would be:
Chemical Kinetics
Solution:
Correct answer is (c) $\frac{0.693}{\sqrt{2k}}$