Q.
When a system is taken from state 1 to 2 along the path $1 a 2$ it absorbs $50\, cal$ of heat and work done is $20\, cal$. Along the path $1 b 2,\, Q=36\, cal$. What is the work done along $1 b 2 ?$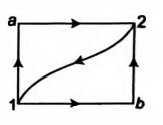
Thermodynamics
Solution: