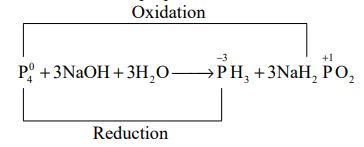
Q. The reaction of white phosphorous with aqueous $NaOH$ gives phosphine along with another phosphorous containing compound. The reaction type; the oxidation states of phosphorus in phosphine and the other product are respectively
AIEEEAIEEE 2012
Solution: