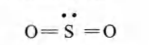Q. The hybridisation of sulphur in sulphur dioxide is
IIT JEEIIT JEE 1986
Solution:
In $SO_2$, the Lewis-dot structure is
Electron pairs at S = 2 ($\sigma$bonds) + 1 (lone-pair) = 3
$sp^2$ hybridised.
NOTE
$\pi$-bonded electrons are not present in hybrid orbitals, therefore not counted in electron pairs. Rather $\pi$ bonds are formed by lateral overlapping of pure p-orbitals.