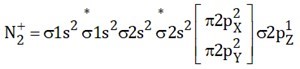Q. $N_{2}$ and $O_{2}$ are converted to mono cations $N_{2}^{+}$ and $O_{2}^{+}$ respectively. The wrong statement with respect to given changes in the following is
NTA AbhyasNTA Abhyas 2020Chemical Bonding and Molecular Structure
Solution: