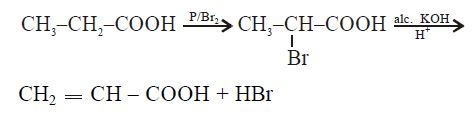Q.
In the following reaction order, B is
$CH_3 CH_2 - COOH \xrightarrow{P/Br_2 }A \xrightarrow[H^+]{alc. KOH} B$
Solution:
$P + Br_2$ is brominating agent that brominates the $\alpha$-position. The product then undergo dehydrohalogenation in the presence of alcoholic potassium hydroxide.