Q.
In a $Cu$-voltameter, mass deposited in $30s$ is $m \,gm$. If the time-current graph is shown in the following figure
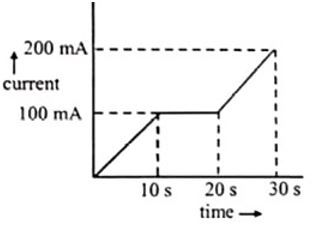
What is the electrochemical equivalent of $Cu$?
NTA AbhyasNTA Abhyas 2022
Solution: