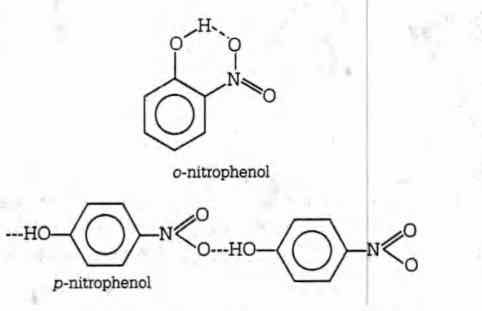
Q. The boiling point of p-nitrophenol is higher than that of o-nitrophenol because
Solution:
The boiling point of p-nitrophenol is higher than that of o-nitrophenol because p-nitrophenol have intermolecular hydrogen bonding whereas o-nitrophenol have intramolecular H-bonding as given below