Q.
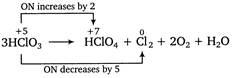
In the disproportionation reaction
the equivalent mass of the oxidizing agent is (molar mass of )
Solution:
This is a disproportionation reaction since here the oxidation state of chlorine decreases from +5 to 0 in as well as increases from +5 to +7 in . Thus, acts as oxidizing as well as reducing agent. Equivalent mass of oxidizing agent (i.e., )