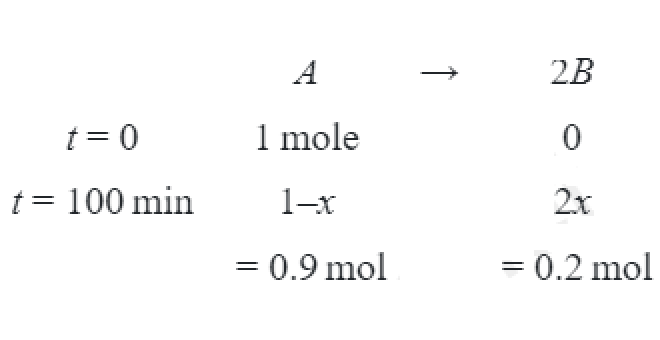- Tardigrade
- Question
- Chemistry
- For the first order reaction A arrow 2 B, 1 mole of reactant A gives 0.2 moles of B after 100 minutes. The half life of the reaction is ...... min. (Round off to the nearest integer). [Use: ln 2=0.69, ln 10=2.3 Properties of logarithms: ln xy=y ln x; . ln ((x/y))= ln x- ln y ] (Round off to the nearest integer)
Q.
For the first order reaction mole of reactant gives moles of after minutes. The half life of the reaction is ...... min.
(Round off to the nearest integer).
[Use : Properties of logarithms : ]
(Round off to the nearest integer)
Answer: 300
Solution: