Q.
For an ideal gas, consider only work on going from initial state to final state . The final state can be reached by either of two paths as shown below. Which of the option is correct? (Take as change in entropy and was work done.)
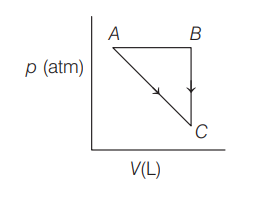
Solution: