- Tardigrade
- Question
- Chemistry
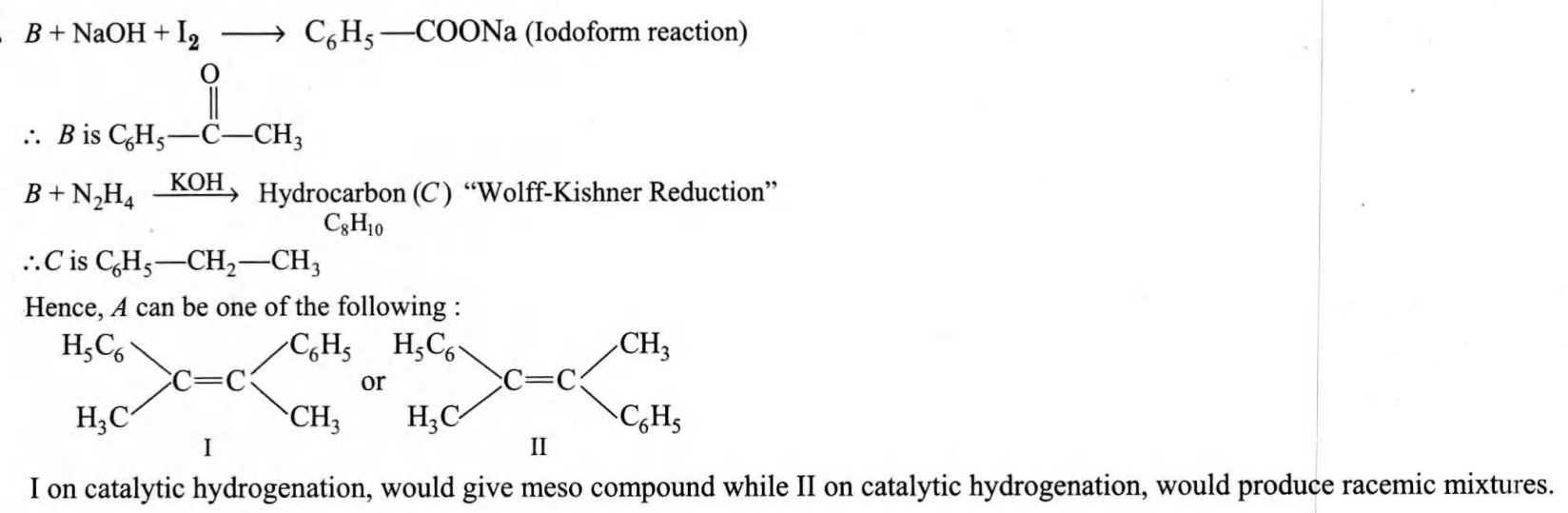
- An alkene (A) C 16 H16 on ozonolysis gives only one product (B) C 8 H 8 O. Compound (B) on reaction with NaOH / I 2 yields sodium benzoate. Compound ( B ) reacts with KOH / NH 2 NH 2 yielding a hydrocarbon (C) C 8 H 10. Write the structures of compounds (B) and (C). Based on this information two isomeric structures can be proposed for alkene (A). Write their structures and identify the isomer which on catalytic hydrogenation ( H 2 / Pd - C) gives a racemic mixture.
Q. An alkene (A) on ozonolysis gives only one product (B) . Compound (B) on reaction with yields sodium benzoate. Compound reacts with yielding a hydrocarbon (C) . Write the structures of compounds (B) and (C). Based on this information two isomeric structures can be proposed for alkene (A). Write their structures and identify the isomer which on catalytic hydrogenation ( ) gives a racemic mixture.
Solution: