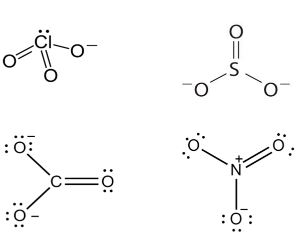
Q. Which of the following pairs of ions are isoelectronic and isostructural ?
AIPMTAIPMT 2015
Solution:
Number of electrons
$\ce{ CO^{2-}_3 = 6 + 2 + 24 = 32}$
$\ce{SO^{2-}_3 = 16 + 2 + 24 = 42}$
$\ce{CIO^{-}_3 = 17 + 24 + 1 = 42}$
$\ce{CO^{2-}_3 = 6 + 24 + 2 = 32}$
$\ce{NO^{2-}_3 = 7 + 2 + 24 = 33 }$
Hence, $\ce{CIO^{-}_3}$ and $\ce{SO^{2-}_3}$ are isoelectronic and are pyramidal in shape.