Q.
Which of the following is true about the complex $\left[ PtCl _{2}\left( NH _{3}\right)\left( OH _{2}\right)\right] ?$ [Atomic number of $Pt =78$ ]
(i) It will have two geometrical isomerić forms, cis and trans
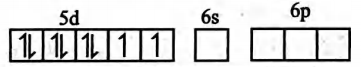
(ii) The hybridisation state of $\operatorname{Pt}( II )$ is $sp ^{3}$
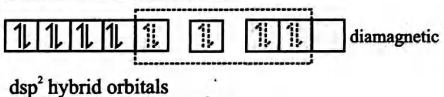
(iii)It is a square planar complex
(iv) It is a diamagnetic complex
(v)It can show hydrate isomerism
(vi) It is a tetrahedral complex
Coordination Compounds
Solution: