Q. Which of the following compounds will give a yellow precipitate with iodine and alkali? (1) 2-hydroxy propane (2) Acetophenone (3) Methyl acetate (4) Acetamide
BHUBHU 2008
Solution:
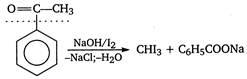
A compound which contains either $ C{{H}_{3}}\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{CH}}\, $ or $ C{{H}_{3}}\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\, $ group, gives yellow precipitate of iodoform with $ {{I}_{2}} $ and alkali. $ \underset{2-hydroxy\text{ }propane}{\mathop{C{{H}_{3}}-\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{3}}}}\,\xrightarrow[-NaCl,-{{H}_{2}}O]{NaOH/{{I}_{2}}} $ $ \underset{iodoform}{\mathop{CH{{I}_{3}}}}\,+C{{H}_{3}}COONa $
$ Methyl\text{ }acetate\xrightarrow[{}]{NaOH/{{I}_{2}}}No\text{ }reaction $ $ Acetamide\xrightarrow{NaOH/{{I}_{2}}}No\text{ }ppt $
$ \therefore $ Only 2-hydroxy propane and acetophenone give positive iodoform test.