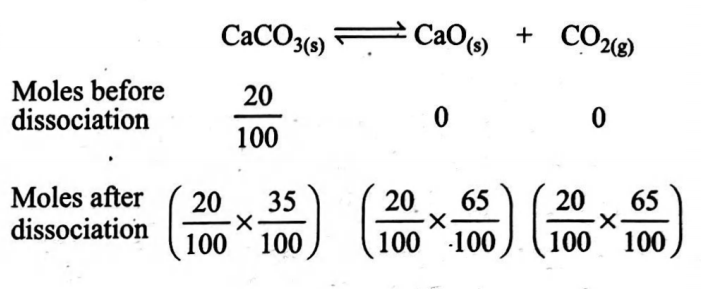Q. When $20\, g$ of $CaCO _{3}$ were put into $10\, L$ flask and heated to $800^{\circ} C , 35 \%$ of $CaCO _{3}$ remained unreacted at equilibrium, $K_{P}$ for decomposition of $CaCO _{3}$ is
Equilibrium
Solution: