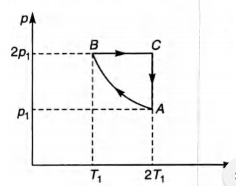
Q.
Two moles of an ideal monoatomic gas is taken through a
cycle ABCA as shown in thep-T diagram. During the process
AB, pressure and temperature of the gas vary such that
pT = constant. If $T_1$ = 300 K, calculate
(a) the work done on the gas in the process AB and
(b) the heat absorbed or released by the gas in each of the
processes.
Give answers in terms of the gas constant R.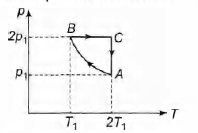
IIT JEEIIT JEE 2000Thermodynamics
Solution:
(a) Number of moles, n = 2,T$_1$ = 300 K
During the process A $\rightarrow$ B
pT = constant or p$^2$V = constant = K (say)
$\therefore \, \, \, \, \, \, \, p=\frac{\sqrt K}{\sqrt V}$
$\therefore \, \, \, \, W_{A \rightarrow \, B} = \int_{VA}^{VB} p.dv=\int_{VA}^{VB}\frac{\sqrt K}{\sqrt V} dV$
$ \, \, \, \, \, \, \, =2\sqrt K [\sqrt KV_B -\sqrt V_A]$
$ \, \, \, \, \, \, \, =2[\sqrt KV_B-\sqrt KV_A]$
$ \, \, \, \, \, \, \, =2[]\sqrt{(p_B^2 V_B)V_B)}-\sqrt{(p_A^2V_A)V_A)}]$
$ \, \, \, \, \, \, \, =2[p_BV_B-p_AV_A]$
$ \, \, \, \, \, \, \, =2[nRT_B-nRT_A]=2nR[T_1-2T_1]$
$ \, \, \, \, \, \, \, =(2)(2))(R)[300-600]=-1200 R$
$\therefore \, $ Work done on the gas in the processes is 1200 R
Alternate solution
$ pV=nRT$
$\therefore \, \, \, \, \, \, \, \, \, \, pdV+Vdp=nRdT$
or $ \, \, \, \, \, \, \, \, \, \, pdV+\frac{(nRT)}{p}.dp=nRdT \, \, \, \, \, \, \, \, $ ....(i)
From the given condition
pT = constant
pdT + Tdp = 0 $\, \, \, \, \, \, \, \, \, \, \, \, \, \, \, \, $... (ii)
From Eqs. (i) and (ii), we get
pdV = 2nRdT
$\therefore \, \, \, \, \, W_{A \rightarrow B}=\int pdV=2nR \int_{T_A}^{T_B}dT=2nR(T_B-T_A)$
$ \, \, \, \, \, \, =2nR(T_1-2T_1)=(2)(2)(R)(300-600)$
$\, \, \, \, \, or \, \, \, \, W_{A \rightarrow \, B}= -1200 R$
(b) Heat absorbed/released in different processes.
Since, the gas is monoatomic.
Therefore, $C_v=\frac{3}{2}R \, and \, C_p =\frac{5}{2}R \, and \, \gamma=\frac{5}{3}$
Process $A \rightarrow \, B \, \, \Delta U =^n C_v \Delta T$
$ \, \, \, \, \, \, \, \, \, =(2)\bigg(\frac{3}{2}R\bigg)(T_B-T_A) $
$ \, \, \, \, \, \, \, \, \, =(2)\bigg(\frac{3}{2}R\bigg)(300-600)=-900 R$
$\therefore \, \, \, \, \, \, \, \, \, Q_{A \rightarrow B}=W_{A \rightarrow B}+\Delta U$
$ \, \, \, \, \, \, \, \, \, \, \, \, \, \, \, \, =(-1200 R)-(900 R)$
$ \, \, \, \, \, \, \, Q_{A \rightarrow B}=-2100 R \, \, \, \, \, \, \, \, \, $ (released)
Alternate solution
In the process pV$^x$ = constant
Molar heat capacity, C = $\frac{R}{\gamma -1}+\frac{R}{1- x}$
Here the process is p$^2$V = constant
or pV$^{ l/2}$ = constant
i.e.,, $ \, \, \, \, \, \, \, \, x=\frac{1}{2}$
$\therefore \, \, \, \, \, \, \, \, C=\frac{R}{\frac{5}{3}-1}+\frac{R}{1-\frac{1}{2}}$
$\therefore \, \, \, \, \, \, \, \, C=3.5 R$
$\therefore \, \, \, \, \, \, \, \, Q_{A \rightarrow B=nC \Delta T}$
$ \, \, \, \, \, \, \, \, \, \, \, \, \, \, \, \, =(2)(3.5 R)(300-600)$
or $ \, \, \, \, \, \, Q_{A \rightarrow B}=-2100 R$
Process B $\rightarrow $ C Process is isobaric
$\therefore \, \, \, \, \, \, \, \, Q_{B \rightarrow C}=nC_p \Delta T$
$ =(2)\bigg(\frac{5'}{2}R\bigg)(T_C -T_B)$
$ =(2)\bigg(\frac{5}{2}R\bigg)(2T_1 -T_1)$
$ = (5R)(600-300)$
$ Q_{B \rightarrow C}=1500 R$ (absorbed)
Process C $\rightarrow $ A Process is isothermal
$\therefore \, \, \, \, \, \, \, \, \, \, \, \, \, \Delta U =0$
and $Q_{C \rightarrow A}=W_{C \rightarrow A} =nRT_C ln \bigg(\frac{P_C}{P_A}\bigg)$
$ =nR(2T_1)ln \bigg(\frac{2p_1}{p_1}\bigg)$
$ =(2)(R)(600)ln (2)$
$Q_{C \rightarrow A =831.6 R}$(absorbed)
NOTE
$\bullet$ In first law of thermodynamics, (dQ = dU + dW) we come across
three terms dQ, dU and dW.
dU = nC$_v$dT for all the processes whether it is isobaric, isochoric or
else and dQ = nCdT where
$ \, \, \, \, \, \, \, \, \, \, C=\frac{R}{\gamma -1}+\frac{R}{1-x}$
in the process pV$^x$ = constant.
$\bullet$ In both the terms we require $dT (=T_1-T_1)$ only. The third term dW is
obviously dQ - dU. Therefore if in any process change in
temperature (dT) and p-V relation is known, then the above method
is the simplest one. Note that even if we have V-T or T-p relation, it
can be converted into p-V relation by the equation
pV = nRT