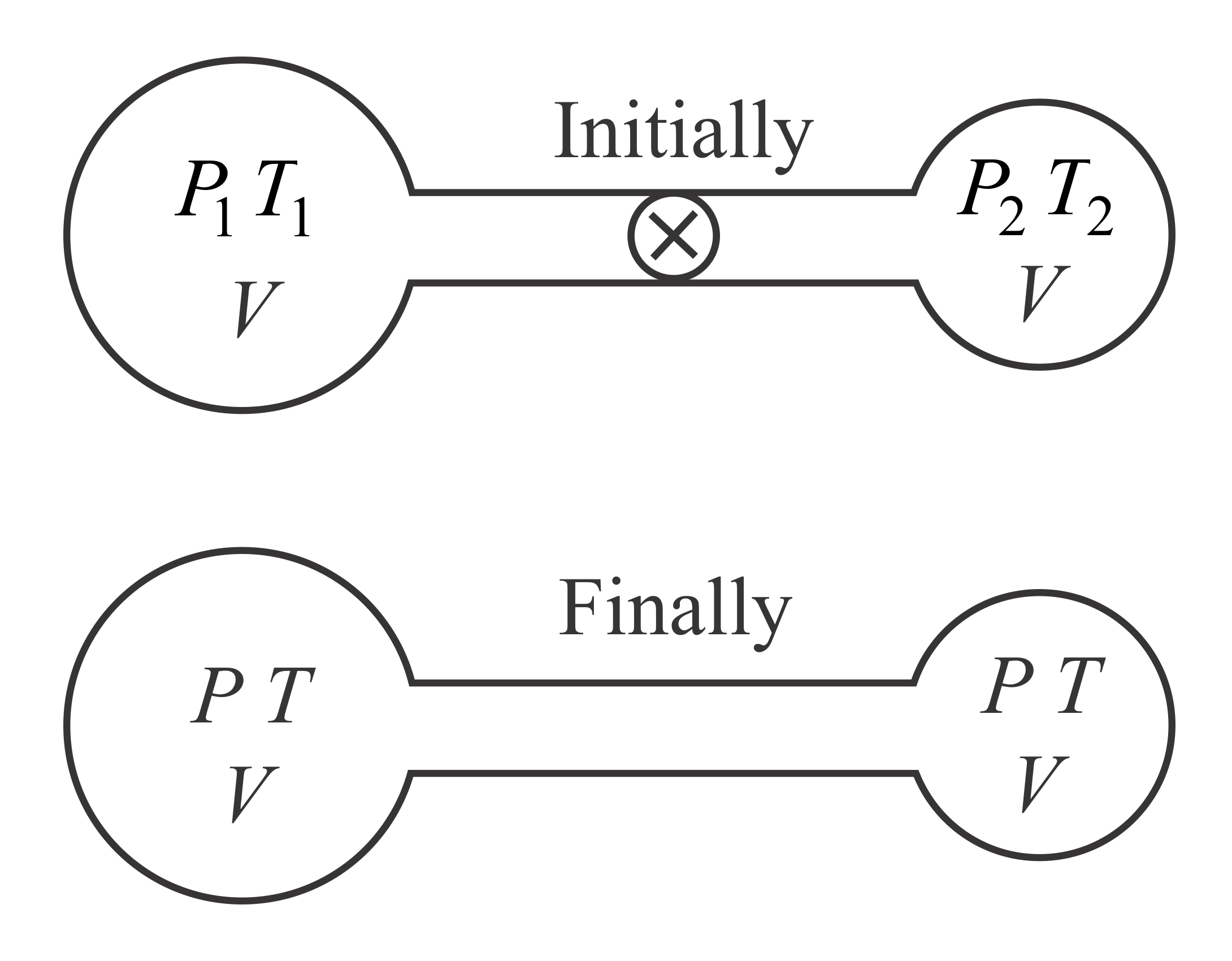
Q. Two containers of the equal volume contain the same gas at pressure $P_{1}$ and $P_{2}$ and absolute temperatures $T_{1}$ and $T_{2}$ , respectively. On joining the vessels, the gas reaches a common pressure $P$ and common temperature $T$ . The ratio $\frac{P}{T}$ is equal to
NTA AbhyasNTA Abhyas 2022
Solution: