Q.
The variation of compressibility factor (Z) with pressure $(p$ in bar) for some gases are shown in the figure below. Identify the gases $(A),(B)$ and $(C)$ respectively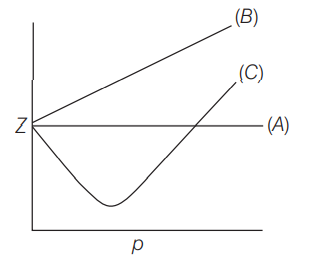
TS EAMCET 2019
Solution: