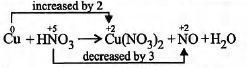Q.
The stoichiometric constants for the reaction
$pCu + qHNO_3 \to rCu(NO_3)_2 + sNO + tH_2O$
$p$, $q$, $r$, $s$ and $t$ respectively are
Redox Reactions
Solution: