Q. The pair in which both the species have the same magnetic moment (spin only) is :
Solution:
Complex
$e^-$ configuration
no. of unpaired $e^-$
$[Mn(H_2O)_6]^{2+}$
WFL
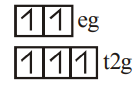
5
$[Cr(H_2O)_6]^{2+}$
WFL
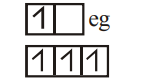
4
$[COCl_4]^{2-}$
Tetrahedral
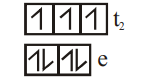
3
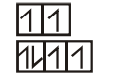
$[Fe(H_2O)_6]^{2+}$
WFL
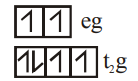
4
$[Co(OH)_4]^{2-}$
WFL
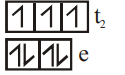
3
Tetrahedral
$[Fe(NH_3)_6]^{2+}$
4
Thus complex $\left[ Cr \left( H _{2} O \right)_{6}\right]^{2+}$ and $\left[ Fe \left( H _{2} O \right)_{6}\right]^{2+}$ have same no. of unpaired $e ^{-}$ and hence same
magnetic moment (spin only).
| Complex | $e^-$ configuration | no. of unpaired $e^-$ |
|---|---|---|
| $[Mn(H_2O)_6]^{2+}$ WFL |
 |
5 |
| $[Cr(H_2O)_6]^{2+}$ WFL |
 |
4 |
| $[COCl_4]^{2-}$ Tetrahedral |
 |
3 |
| $[Fe(H_2O)_6]^{2+}$ WFL |
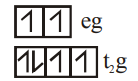 |
4 |
| $[Co(OH)_4]^{2-}$ WFL |
 |
3 |
| Tetrahedral $[Fe(NH_3)_6]^{2+}$ |
 |
4 |