Q. The main difference in $C — X$ bond of a haloalkane and a haloarene is
Haloalkanes and Haloarenes
Solution:
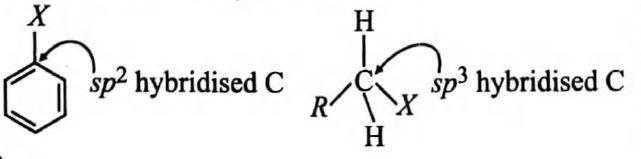
In haloarenes, carbon of $C—X$ is $sp^2$ hybridised while in haloalkanes it is $sp^3$ hybridised.
Solution:
