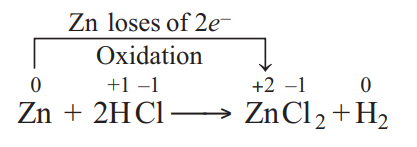Q.
Identify the correct statement in relation to the following reaction.
$Zn +2 HCl \longrightarrow ZnCl _{2}+ H _{2}$
Redox Reactions
Solution:
Solution: