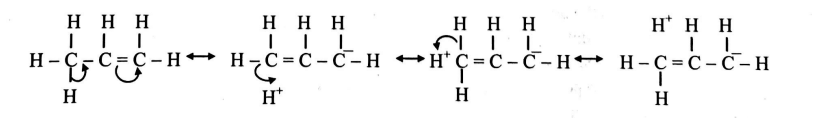
Q. How many no-bond resonating structures can be drawn for alkene having $C _{3} H _{6}$ formula?
Hydrocarbons
Solution:
No. of hyperconjugation structures $=$ No. of $\alpha-H$ atoms adjacent to $sp ^{2}$ hybridized $C$-atom
Three $\alpha$-hydrogens are present in propene $\left( C _{3} H _{6}\right)$. Thus, three no-bond resonating structures (or hyperconjugation structures) can be drawn for propene.