Q.
Graphs below show the entropy versus energy $U$ of
two systems $1$ and $2$ at constant volume. The initial
energies of the systems are indicated by $U_{1,i}$. and
$U_{2,i}$., respectively. Graphs are drawn to the same scale. The systems are the n brought into thermal contact with each other. Assume that, at all times the
combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium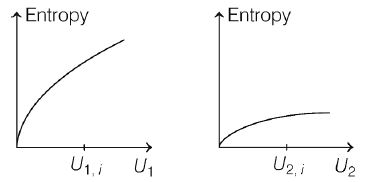
KVPYKVPY 2018
Solution: