Q.
Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, then :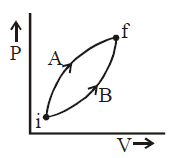
Solution:
Initial and final states for both the processes are
same.
$\therefore \, \, \Delta U_A = \Delta U_B$
Work done during process $A$ is greater than in
process $B$.
By First Law of thermodynamics
$\Delta Q = \Delta U + W$
$\Rightarrow \, \, \Delta Q_A > \Delta Q_B$