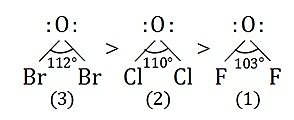Q.
Consider the following oxides
$1.$ $OF_{2}$
$2.$ $Cl_{2}O$
$3.$ $Br_{2}O$
The correct sequence of $X-O-X$ bond angle is
NTA AbhyasNTA Abhyas 2020The p-Block Elements - Part2
Solution: