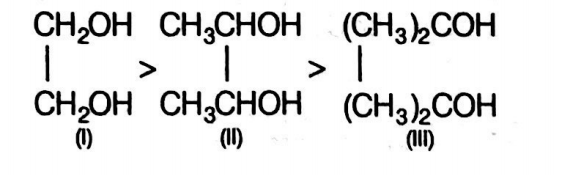Q.
Arrange the following in the order of rate of oxidation with periodic acid
I. $HOCH _{2} CH _{2} OH$,
II. $CH _{3} CHOHCHOHCH _{3}$
III. $(CH \left._{3}\right)_{2} COHCOH \left( CH _{3}\right)_{2}$
Bihar CECEBihar CECE 2013Alcohols Phenols and Ethers
Solution: