Q.
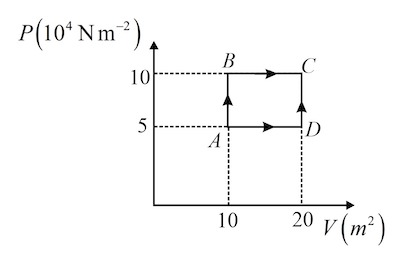
A sample of $2kg$ monoatomic helium gas (assumed ideal) is taken through the process $ABC$ and another sample of $2kg$ of the same gas is taken through the process $ADC$ . Given that the molecular mass of helium = $4amu$ , find the temperature of helium in the state $D$ . $\left[\right.$ Take the universal gas constant $R=\frac{25}{3}Jmol^{- 1}K^{- 1}\left]\right.$
NTA AbhyasNTA Abhyas 2022
Solution: