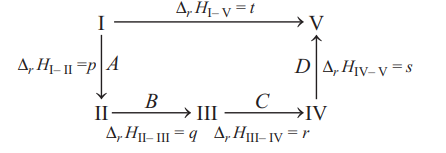Q. A reaction takes place in a several sequential steps $A$, $B, C$ and $D$. The value of enthalpy change for sequential steps are $p, q, r$ and $s$, respectively. If $B$ and $C$ have equal values of enthalpy change with total enthalpy change $t$, then choose the incorrect symbolic equation among the following.
Thermodynamics
Solution: