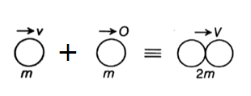Q. A hydrogen atom moving at speed $v$ collides with another hydrogen atom kept at rest. Find the minimum value of $v$ for which one of the atoms may get ionised. The mass of a hydrogen atom $=1.67\times 10^{- 27 \, }kg.$ Assume completely inelastic collision.
NTA AbhyasNTA Abhyas 2020Atoms
Solution: