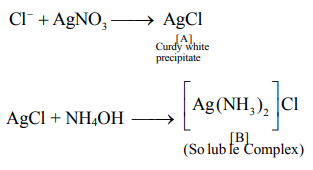
Q. A chloride salt solution acidified with dil. $HNO _3$ gives a curdy white precipitate, $[A]$, on addition of $AgNO _3 - [A]$ on treatment with $NH _4 OH$ gives a clear solution, $B$. $A$ and $B$ are esepectirely
Solution:
Correct answer is (c) $AgCl \&\left[ Ag \left( NH _3\right)_2\right] Cl$