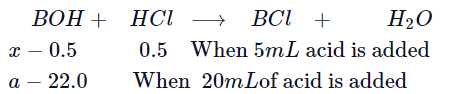Q. A $40 \,ml$. solution of a weak base, $BOH$ is titrated with $0. 1\,N HCl$ solution. The $pH$ of the solution is found to be $10.04$ and $9.14$ after the addition of $5.0 \,mL $ and $20.0 \,mL$ of the acid respectively. Find out the dissociation constant of the base.
IIT JEEIIT JEE 1991Equilibrium
Solution: