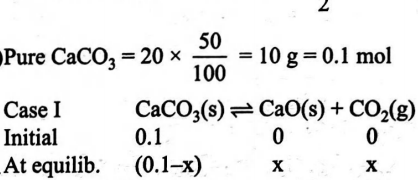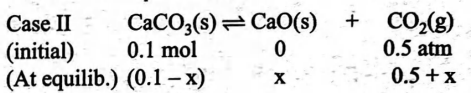Q.
$20\, g$ of sample of $CaCO _{3}( s )$ of $50 \%$ impurity is placed in a sealed vessel of $1\, L$ capacity at $400\, K$ when $50 \% T$ dissociation takes place. If $CO _{2}$ gas at $0.5\, atm$ is already present, percent dissociation of $CaCO _{3}$ is
$CaCO _{3}( s ) \rightleftharpoons CaO ( s )+ CO _{2}( g )$
Equilibrium
Solution: