Q.
$1,3$ -butadiene and $1$-butyne exist in equilibrium in gaseous phase at $298\, K$.
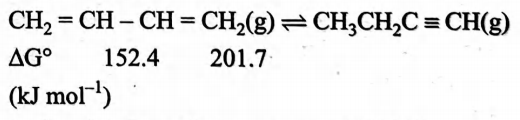
Ratio of their partial pressures at $298\, K$ is
Equilibrium
Solution: