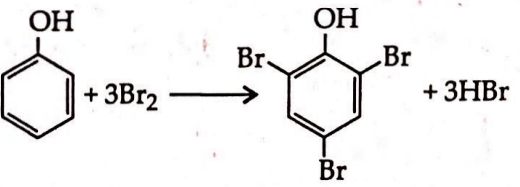
Q. 0.24 mole of phenol was treated with excess of bromine in presence of water. All the phenol got quantitatively converted to bromophenol. The number of moles of $Br _{2}$ reacted were
Alcohols Phenols and Ethers
Solution: