Q.
0.2 moles of an ideal gas is taken round the cycle $ABC$ as shown in the figure. The path $B \rightarrow C$ is an adiabatic process, $A \rightarrow B$ is an isochoric process and $C \rightarrow A$ is an isobaric process. The temperature at $A$ and $B$ are $T_{A}=300\, K$ and $T_{B}=500\, K$ and pressure at $A$ is $1 atm$ and volume at $A$ is 4.9 L. The volume at $C$ is (Given: $\gamma=\frac{C_{P}}{C_{V}}=\frac{5}{3}, R=8.205 \times 10^{-2} L \,atm\, mol ^{-1} K ^{-1}$ $\left(\frac{3}{2}\right)^{2 / 5}=0.81)$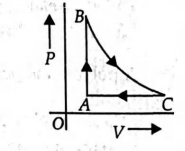
Thermodynamics
Solution: