- Tardigrade
- Question
- Chemistry
- In acid-base titration react rapidly to neutralize each other. Equivalence point is a point at which the acid and the base (or oxidizing agent and reducing agent) have been added in equivalent quantities. The end point is the point at which the titration stops. Since the purpose of the indicator is to stop the titration close to the point at which the acid and base were added in equivalent quantities, it is important that the equivalent point and the end point be as close as possible. The indicator must change colour at a pH close to that of a solution of the salt of the acid and base. Significantly, the pH changes most rapidly near the equivalence point. The exact shape of a titration curve depends on Ka and Kb of acid and base <img class=img-fluid question-image alt=image src=https://cdn.tardigrade.in/img/question/chemistry/3f1a6ef0aa669cc4456b62711ff24a52-.png /> The following curve represents titration curve of HCl against KOH. The pH at equivalent point is Examine the titration curve below and answer the question
Q.
In acid-base titration react rapidly to neutralize each other. Equivalence point is a point at which the acid and the base (or oxidizing agent and reducing agent) have been added in equivalent quantities. The end point is the point at which the titration stops. Since the purpose of the indicator is to stop the titration close to the point at which the acid and base were added in equivalent quantities, it is important that the equivalent point and the end point be as close as possible. The indicator must change colour at a pH close to that of a solution of the salt of the acid and base. Significantly, the pH changes most rapidly near the equivalence point. The exact shape of a titration curve depends on and of acid and base
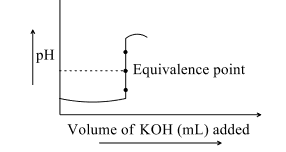
The following curve represents titration curve of against . The at equivalent point is Examine the titration curve below and answer the question
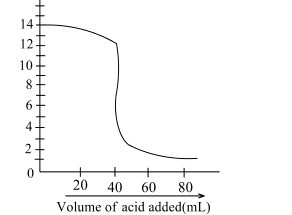
Solution: